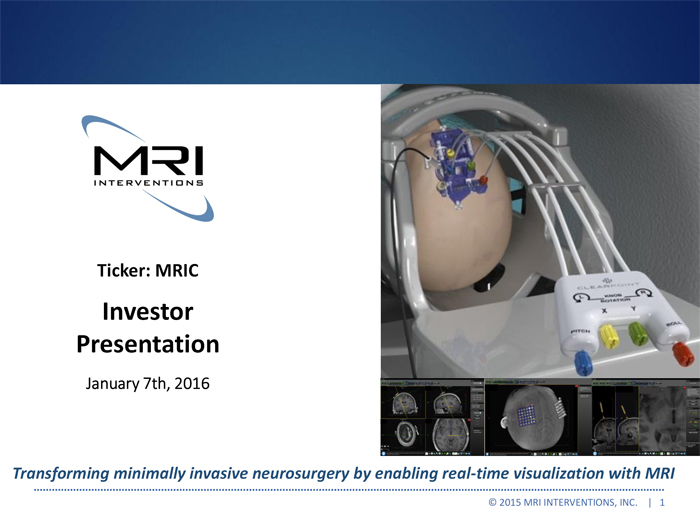
Exhibit 99.2

Ticker: MRIC
Investor Presentation
January 7th, 2016
Transforming minimally invasive neurosurgery by enabling real-time visualization with MRI
c 2015 MRI INTERVENTIONS, INC. | 1

Forward Looking Statements
Certain statements in this presentation may constitute forward-lookingstatements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements often can be identified by words such as “anticipates,” “believes,” “could,” “estimates,” “expects,” “intends,” “may,” “plans,” “potential,” “predicts,” “projects,” “should,” “will,” “would,” or the negative of these words or other words of similar meaning. Forward-looking statements by their nature address matters that, to different degrees, are uncertain and involve risk. Uncertainties and risks may cause MRI Interventions’ actual results and the timing of events to differ materially from those expressed in or implied by MRI Interventions’ forward-looking statements. Particular uncertainties and risks include, among others: demand and market acceptance of our products; our ability to successfully expand, and achieve full productivity from, our sales, clinical support and marketing capabilities; availability and adequacy of reimbursement from third party payors for procedures utilizing our products; the sufficiency of our cash resources to maintain planned commercialization efforts and research and development programs; future actions of the FDA or any other regulatory body that could impact product development, manufacturing or sale; our ability to protect and enforce our intellectual property rights; our dependence on collaboration partners; the impact of competitive products and pricing; the impact of the commercial and credit environment on us and our customers and suppliers; and our ability to successfully complete the development of, and to obtain regulatory clearance or approval for, our ClearTrace system. More detailed information on these and additional factors that could affect MRI Interventions’ actual results and the timing of events are described in its filings with the Securities and Exchange Commission. Except as required by law, MRI Interventionsundertakesnoobligationtopubliclyupdateorreviseanyforward-looking statementsmadeinthispresentationtoreflectanychangeinMRIInterventions’expectationsoranychange inevents,conditionsorcircumstancesonwhichanysuchstatementsarebased.
c 2015 MRI INTERVENTIONS, INC. | 2
MRI Interventions Opportunity
Market potential
Market Potential 55,000 potential ClearPoint procedures across multiple therapies
Electrode placement for deep brain stimulation
NavigationSystem
Laser Ablation for ablation of epileptic foci or Brain Tumors
forMultiple
Brain Tumor Biopsy for deep seated tumors
Therapies
Precise Drug Delivery to target lesions
Opportunityto Area of interest to large medical device companies
AttractMultiple Medtronic, St. Jude and Boston Scientific investing in neuro market Players MRI Scanner Companies embracing MRI-guided therapies
Drug Companies pursuing direct delivery
Focused commercial effort in neurosurgery; FDA/CE approved products
Uniquely Delivery platform for multiple therapies Positioned Established, proprietary IP position
Recent restructuring complete
c 2015 MRI INTERVENTIONS, INC. | 3

MRI Interventions:
Real Time MRI Guided Surgery
Firstto-markettechnologyplatformenabling altime MRI guideded surgery; Acleared, CE-marked and nd40+ ClearPoint sites
Focused commercialization of neuro platform underway; recent restructuring complete
Attractive razor/razorblade business model with strongng potential margins
Compatiblewithall major MRI manufacturers; Interoperability w/ Medtronic, Monteris, neuro roducts
Established intellectual property portfolio
Professionalmanagementteamwithrelevantmedicaldevicecommercialization experience: Intuitive, Medtronic, Kyphon, Boston Scientific, ev3, Edwards Lifescience, Cordis
c 2015 MRI INTERVENTIONS, INC. | 4
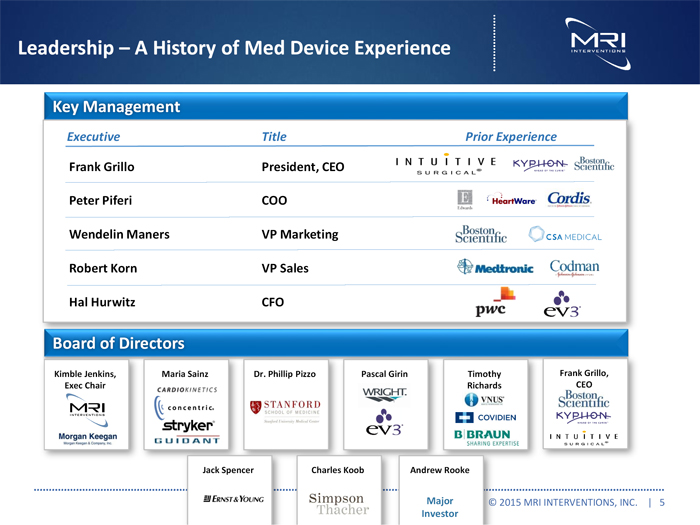
Leadership A History of Med Device Experience
Key Management
Executive Title Prior Experience
Frank Grillo President, CEO
Peter Piferi COO
Wendelin Maners VP Marketing
Robert Korn VP Sales
Hal Hurwitz CFO
Board of Directors
Kimble Jenkins, Maria Sainz Dr. Phillip Pizzo Pascal Girin Timothy Frank Grillo, Exec Chair Richards CEO
Jack Spencer Charles Koob Andrew Rooke
Major 2015 MRI INTERVENTIONS, INC. | 5
Investor
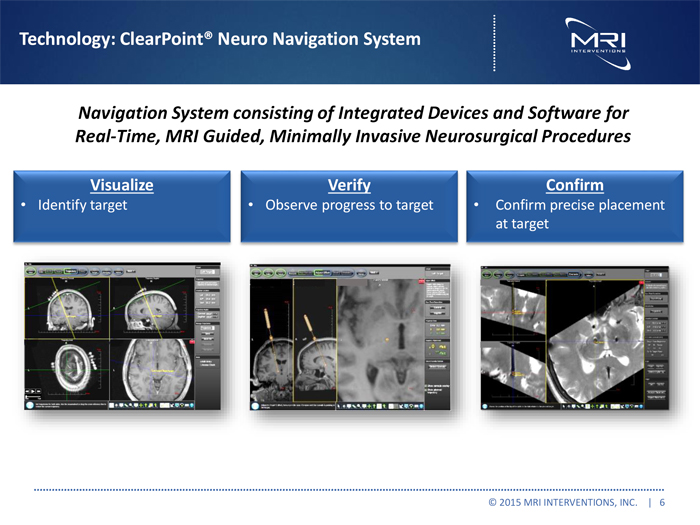
Technology: ClearPointR Neuro Navigation System
Navigation System consisting of Integrated Devices and Software for Real-Time, MRI Guided, Minimally Invasive Neurosurgical Procedures
Visualize Verify Confirm
Identify target Observe progress to target Confirm precise placement at target
c 2015 MRI INTERVENTIONS, INC. | 6
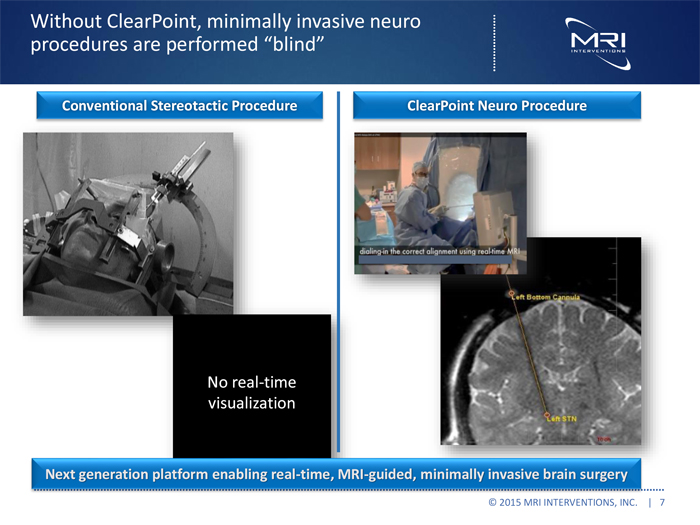
Without ClearPoint, minimally invasive neuro procedures are performed “blind”
Conventional Stereotactic Procedure ClearPointnt Neuro Procedure
No real-time visualization
Nextt generationn platform enabling realtime, MRIIguided, minimally invasive brain surgery
c 2015 MRI INTERVENTIONS, INC. | 7
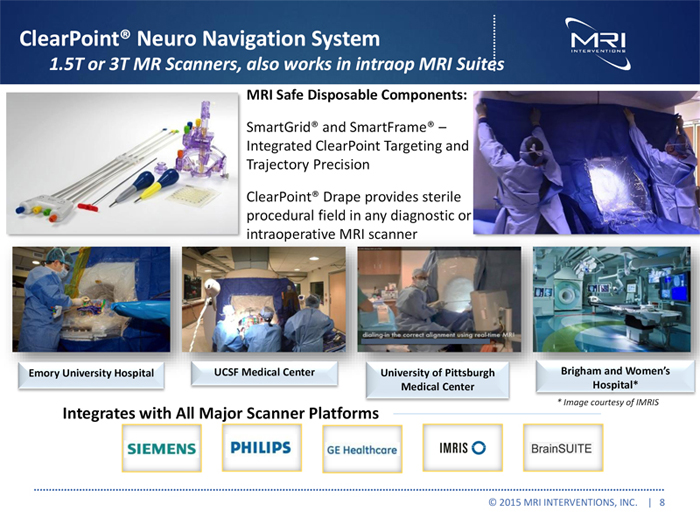
ClearPointR Neuro Navigation System
1.5T or 3T MR Scanners, also works in intraop MRI Suites
MRI Safe Disposable Components:
SmartGridR and SmartFrameR Integrated ClearPoint Targeting and Trajectory Precision
ClearPointR Drape provides sterile procedural field in any diagnostic or intraoperative MRI scanner
Emory University Hospital UCSF Medical Center University of Pittsburgh Brigham and Women’s Medical Center Hospital*
* Image courtesy of IMRIS
Integrates with All Major Scanner Platforms
c 2015 MRI INTERVENTIONS, INC. | 8
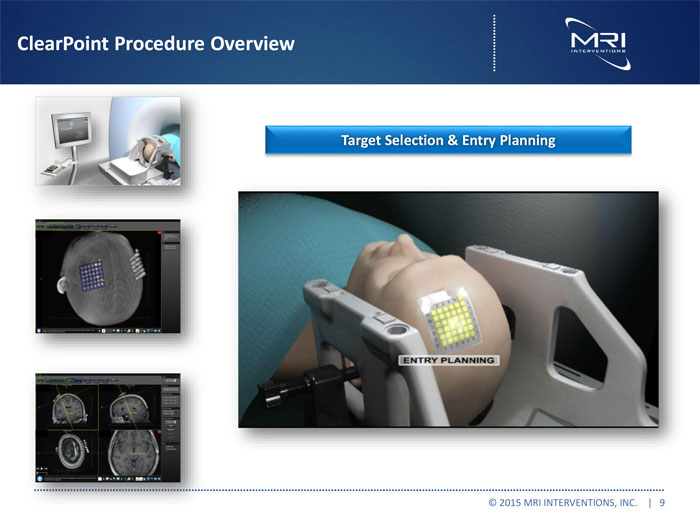
ClearPoint Procedure Overview
Target Selection & Entry Planning
VIDEO
c 2015 MRI INTERVENTIONS, INC. | 9
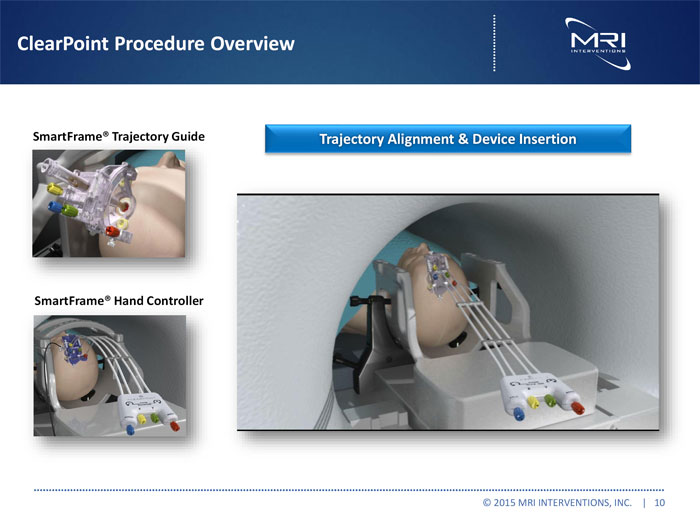
ClearPoint Procedure Overview
SmartFrameR Trajectory Guide Trajectory Alignment & Device Insertion
VIDEO
SmartFrameR Hand Controller
c 2015 MRI INTERVENTIONS, INC. | 10
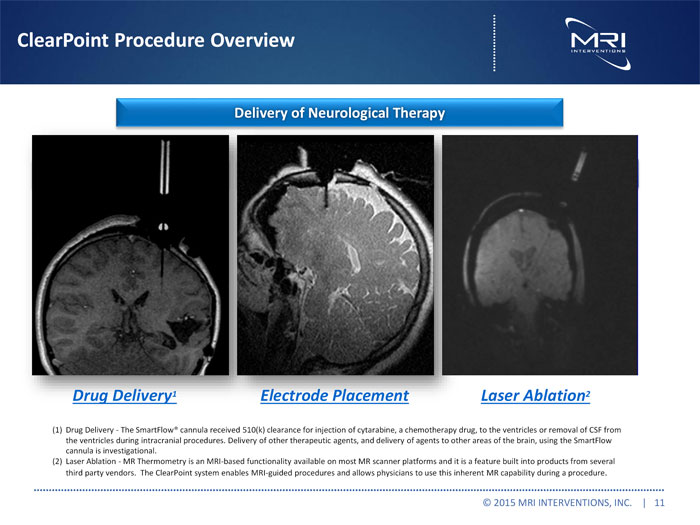
ClearPoint Procedure Overview
Delivery of Neurological Therapy
Drug Deliveryry(1) Electrode Placement Laser Ablationn(2)
VIDEO VIDEO VIDEO
Drug Delivery1 Electrode Placement Laser Ablation2
(1)Drug Delivery -The SmartFlowR cannula received 510(k) clearance for injection of cytarabine, a chemotherapy drug, to the ventricles or removal of CSF from the ventricles during intracranial procedures. Delivery of other therapeutic agents, and delivery of agents to other areas of the brain, using the SmartFlow cannula is investigational.
(2)Laser Ablation -MR Thermometry is an MRI-based functionality available on most MR scanner platforms and it is a feature built into products from several third party vendors. The ClearPoint system enables MRI-guided procedures and allows physicians to use this inherent MR capability during a procedure.
c 2015 MRI INTERVENTIONS, INC. | 11
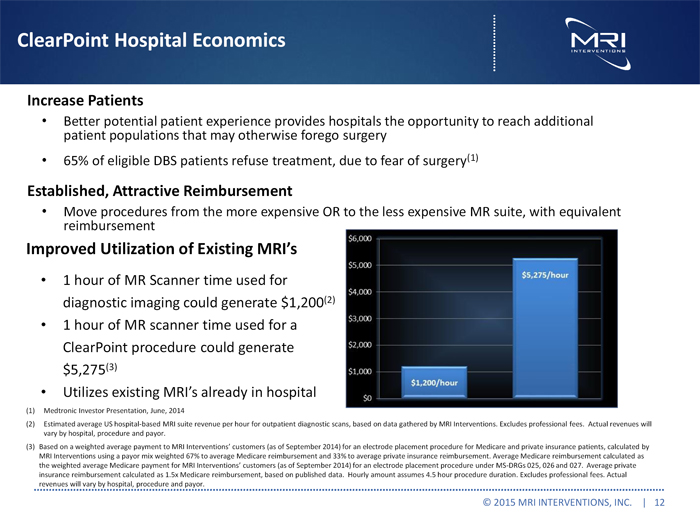
ClearPoint Hospital Economics
Increase Patients
Better potential patient experience provides hospitals the opportunity to reach additional patient populations that may otherwise forego surgery
65% of eligible DBS patients refuse treatment, due to fear of surgery(1)
Established, Attractive Reimbursement
Move procedures from the more expensive OR to the less expensive MR suite, with equivalent reimbursement
Improved Utilization of Existing MRI’s
1 hour of MR Scanner time used for diagnostic imaging could generate $1,200(2)
1 hour of MR scanner time used for a ClearPoint procedure could generate $5,275(3)
Utilizes existing MRI’s already in hospital
(1) Medtronic Investor Presentation, June, 2014
(2) Estimated average US hospital-based MRI suite revenue per hour for outpatient diagnostic scans, based on data gathered by MRI Interventions. Excludes professional fees. Actual revenues will vary by hospital, procedure and payor.
(3) Based on a weighted average payment to MRI Interventions’ customers (as of September 2014) for an electrode placement procedure for Medicare and private insurance patients, calculated by MRI Interventions using a payor mix weighted 67% to average Medicare reimbursement and 33% to average private insurance reimbursement. Average Medicare reimbursement calculated as the weighted average Medicare payment for MRI Interventions’ customers (as of September 2014) for an electrode placement procedure under MS-DRGs 025, 026 and 027. Average private insurance reimbursement calculated as 1.5x Medicare reimbursement, based on published data. Hourly amount assumes 4.5 hour procedure duration. Excludes professional fees. Actual revenues will vary by hospital, procedure and payor.
c 2015 MRI INTERVENTIONS, INC. | 12

Multi-Therapy MRI-Guided Navigational System
Notable Neurosurgeon Supporters
Dr. Philip Starr Dr. Paul Larson Dr. Robert Gross Dr. Robert Wharen, Jr. Dr KrysBankiewicz DrRussLonser ASSFN Past President UCSF & VA Emory University Mayo Clinic - Bankiewicz Lab, UCSF OSU NIH
Jacksonville
Published Peer-Reviewed Journal Support
Compatible With Multiple Therapies
VisualaseR
SmartFlow™ cannula for local drug delivery
c 2015 MRI INTERVENTIONS, INC. | 13

Patented Intellectual Property
Close to 100 issued patents around the world
40+ U.S. 50+ OUS 50+ U.S. Patents 45+ OUS Patents Patent Applications Patent Applications
Issued patents cover areas such as: MRI-guided surgical systems that include software and devices; the SmartFrameR trajectory guide; other ClearPointR disposable components; active intracranial probes; MRI-compatible catheters; MRI-safety technology
Key ClearPoint-related patents do not begin to expire until 2027
c 2015 MRI INTERVENTIONS, INC. | 14

Patient Impact
Martin’s Story
ClearPoint-Enabled Electrode Placement
VIDEO
c 2015 MRI INTERVENTIONS, INC. | 15
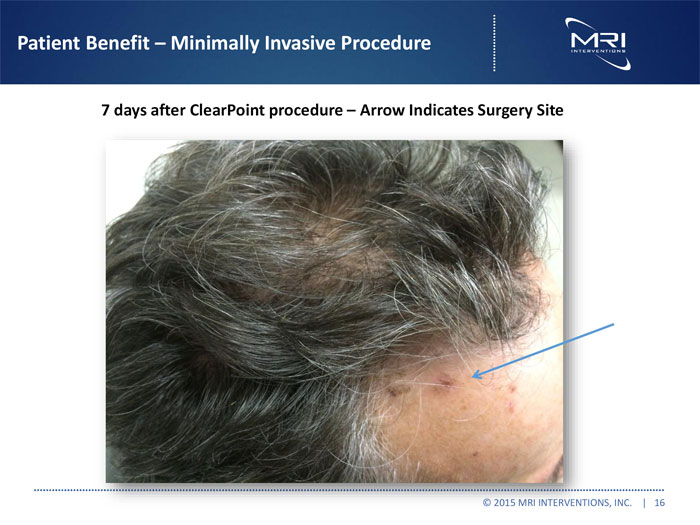
Patient Benefit Minimally Invasive Procedure
7days after ClearPoint procedure Arrow Indicates Surgery Site
ERVENTI INC. | 16
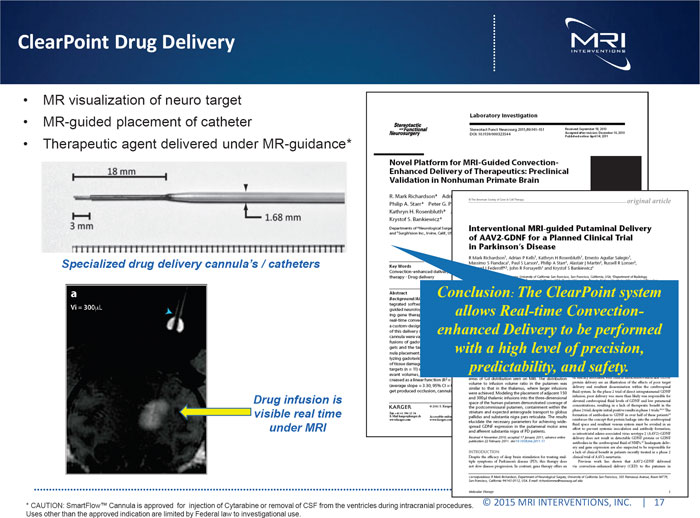
ClearPoint Drug Delivery
MR visualization of neuro target
MR-guided placement of catheter
Therapeutic agent delivered under MR-guidance*
Specialized drug delivery cannula’s / catheters
Conclusion: The ClearPoint system allows Real-time Convection-enhanced Delivery to be performed with a high level of precision, predictability, and safety.
Drug infusion is visible real time under MRI
* CAUTION: SmartFlow Cannula is approved for injection of Cytarabine or removal of CSF from the ventricles during intracranial procedures. c 2015 MRI INTERVENTIONS, INC. | 17

Uses other than the approved indication are limited by Federal law to investigational use.
MRIC’s Unique Opportunity for Drug Delivery
Major Challenges in Delivering Drugs to the Brain
- Blood brain barrier blocks systemic delivery of almost all drugs up to 98% of small molecules
- Direct injection without ClearPoint is blind, so target can frequently be missed
- Neopharm Trial-51% of 572 catheters failed to meet all positioning criteria
Major Benefits of Drug Delivery with ClearPoint
- Neurosurgeon can see that target is reached
- Eliminates the blood brain barrier issue; Reduces/eliminates unwanted systemic side effects; Reduces dosage levels(as little as 1/300th of systemic volumes)
Business Model MRIC Partners with Drug Companies and Researchers
- MRIC provides ClearPoint; Drug company provides drug candidate
- Drug company/sponsor pays for trial
- If drug is approved, MRIC gets device revs (~$7000/case); Drug co gets drug revs
c 2015 MRI INTERVENTIONS, INC. | 18

The ClearPoint Difference
Without ClearPoint (Stereotactic) With ClearPoint
Nodirectvisualization;Performedinanoperating Directhighresolutionvisualization;Performedin room an MRI Suite
Accuracy to target based on prior images Highly accurate, based on real time images Patient may be awake forown brain surgery(1) Patient may be undergeneralanesthesia(1) Long OR time for the patient Can be up to 8 hours Shorterprocedures Can be 3 hours or less
May require OR and MRI for same procedure (laser MRI only procedure -One procedure, one place ablation)
Poor economics for hospital and physician Attractive economics for hospital and physician
Better for Patients Better for Surgeons Better for Hospitals
(1) Microelectrode recording and macrostim are processes that involve listening to neuronal firings (i.e., physiological recordings) and observing physiological responses to stimuli during brain surgery. In connection with our 510(k) clearance in 2010, the FDA requested a warning within ClearPoint’s Instructions for Use based on the lack of data with respect to deep brain stimulation (DBS) procedures. The warning states that the ClearPoint system, alone, should not be usedtoguide a DBS lead to a specific brain target and that final placement of DBS leads requires physiological recordings to confirm that they are located in the correct brain targetand functioning as intended.
c 2015 MRI INTERVENTIONS, INC. | 19

ClearPoint Revenue Model
BUSINESS MODEL RAZOR / RAZORBLADE
ClearPoint Hardware/Software: $100,000 -$150,000 ASP
ClearPoint Disposables: $7,500 (average) ASP per procedure with potentially strong margins Recurring revenue from the sale of disposables Procedures covered by existing inpatient DRG reimbursement codes
c2015MRIINTERVENTIONSINC. 20
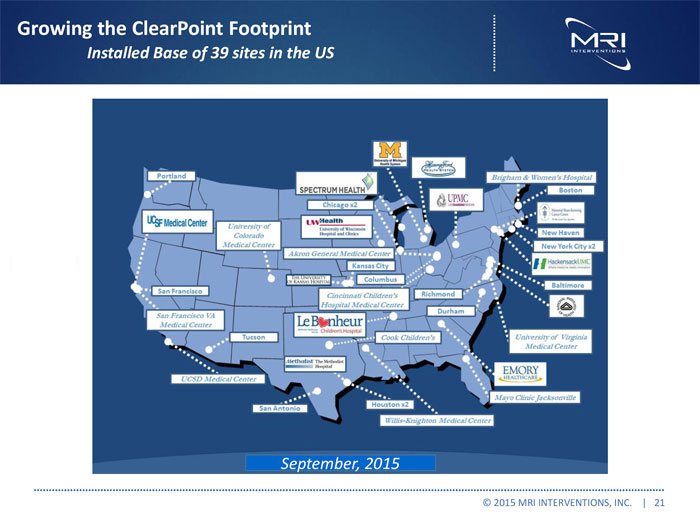
Growing the ClearPoint Footprint
Installed Base of 39 sites in the US
September, 2015
c 2015 MRI INTERVENTIONS, INC. | 21

ClearPoint US Market Opportunity
Parkinson’s Epilepsy Brain Tumors
Total Prevalence (US) 1,500,000 2,200,000 80,000 (annual diagnosis)
Prevalence Drug Treatment 125,000 264,000 Resistant (DTR)
7,500 18,000 Resections: 80,000
Incidence DTR
Stereotactic Biopsy: 10,000
Electrode Laser Ablation Biopsy / Laser Ablation /
ClearPoint Enabled Therapy
Placement (DBS) RNS1 Drug Delivery
Potential ClearPoint 12,500 28,000 14,500 Procedures, Annually2
55,000+ Potential Procedures Per Year
Note: Prevalence and Incidence based on either market research conducted by a third party on behalf of MRI Interventions or researchconducted by MRI Interventions of publicly available sources.
(1) Responsive neurostimulation device (RNS)
(2) Potential Annual ClearPoint Procedures based upon 5% of prevalence and 85% of incidence; Potential Annual ClearPoint Procedures for brain tumors based on market research conducted by a third party on behalf of MRI Interventions.
c 2015 MRI INTERVENTIONS, INC. | 22
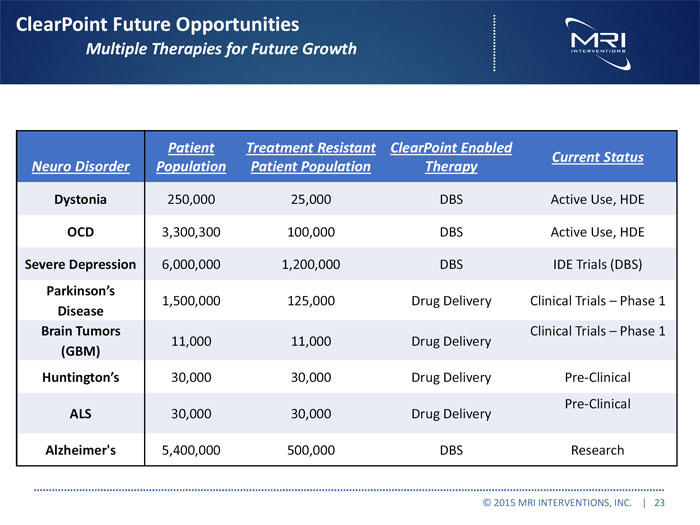
ClearPoint Future Opportunities
Multiple Therapies for Future Growth
Patient Treatment Resistant ClearPoint Enabled
Current Status Neuro Disorder Population Patient Population Therapy
Dystonia 250,000 25,000 DBS Active Use, HDE
OCD 3,300,300 100,000 DBS Active Use, HDE
SevereDepression 6,000,000 1,200,000 DBS IDE Trials (DBS)
Parkinson’s
1,500,000 125,000 Drug Delivery Clinical Trials Phase 1
Disease
Brain Tumors Clinical Trials Phase 1 11,000 11,000 Drug Delivery
(GBM)
Huntington’s 30,000 30,000 Drug Delivery Pre-Clinical
Pre-Clinical ALS 30,000 30,000 Drug Delivery
Alzheimer’s 5,400,000 500,000 DBS Research
c 2015 MRI INTERVENTIONS, INC. | 23
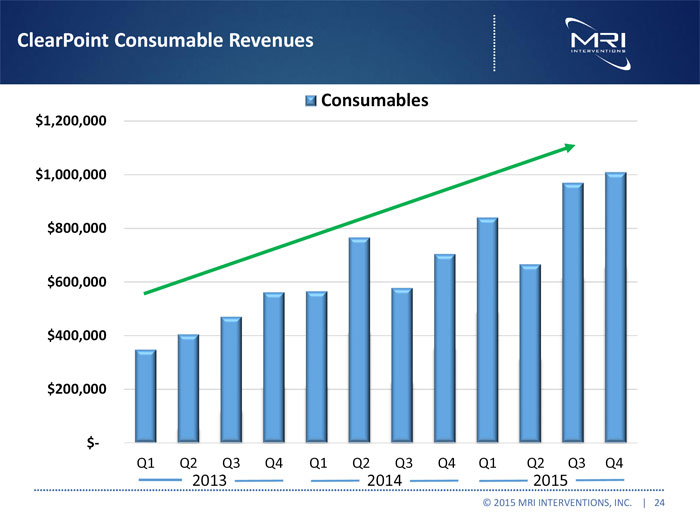
ClearPoint Consumable Revenues
Consumables
$1,200,000 $1,000,000 $800,000 $600,000 $400,000 $200,000
$-
Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4
2013 2014 2015
c 2015 MRI INTERVENTIONS, INC. | 24
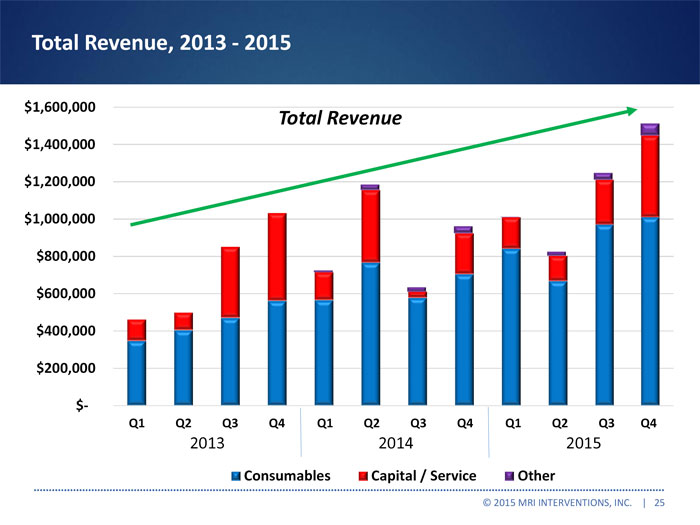
Total Revenue, 2013 -2015
$1,600,000
Total Revenue
$1,400,000 $1,200,000 $1,000,000 $800,000 $600,000 $400,000 $200,000
$-
Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4 Q1 Q2 Q3 Q4
2013 2014 2015
Consumables Capital / Service Other
c 2015 MRI INTERVENTIONS, INC. | 25
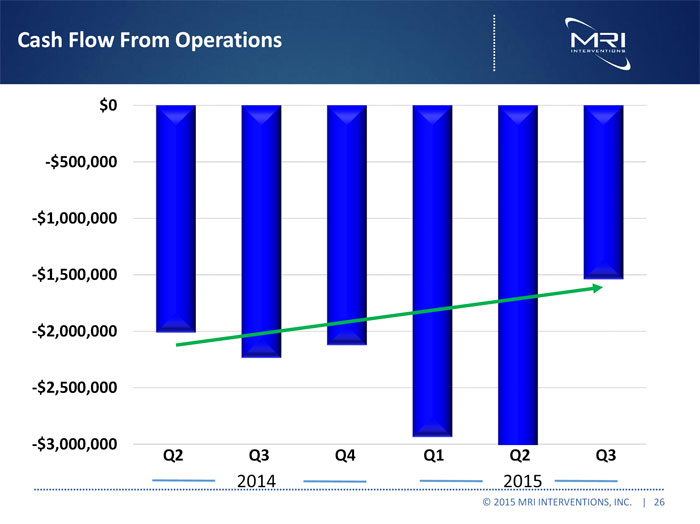
Cash Flow From Operations
$0 -$500,000 -$1,000,000 -$1,500,000 -$2,000,000
-$2,500,000
-$3,000,000
Q2 Q3 Q4 Q1 Q2 Q3
2014 2015
© 2015 MRI INTERVENTIONS, INC. | 26

Commercial Priorities
Focus on adding surgeons at existing accounts
ncrease
Target high volume sites, including epilepsy and tumor neurosurgeons within each Utilization account; gain greater share of their procedures
Add Clinical Specialists and sales reps to commercial team; compensate for utilization growth
Increase peer-to-peer events, presence at trade shows
Enhance Highlight existing data on ClearPoint applications to neurologists and neurosurgeons Communication Communicate value proposition across procedures:
Accuracy
Real time visualization
Improved workflow
Increase patient volume
Identify highest volume potential accounts across multiple procedures
ExpandAccountpand Acco
Support local hospital marketing efforts
Base Capitalize on interest in drug delivery to expand in oncology accounts
Add sales reps
Tightly control working capital, consumption of cash
Achieve Cash
Hire additional personnel only in key functions commercial team; engineering
Breakeven
c 2015 MRI INTERVENTIONS, INC. | 27

R&D Priorities
Seek significant upgrade to existing software; includes Software 2.0 real time fusion, enhanced graphic and User Interface
Technology licenses near complete with three additional software companies for this effort
Through partnership(s), expand our products OR SmartFrame and brand into the operating room for CT based neuro procedures
Establish additional drug delivery partnerships, Drug Delivery and participate in additional clinical trials
Become the neuro drug delivery device partner of choice
Continue to enhance product line with a focus Bolt Driver
Procedure on procedure simplification and consistency for
Enhancements
Laser Ablation
c 2015 MRI INTERVENTIONS, INC. | 28
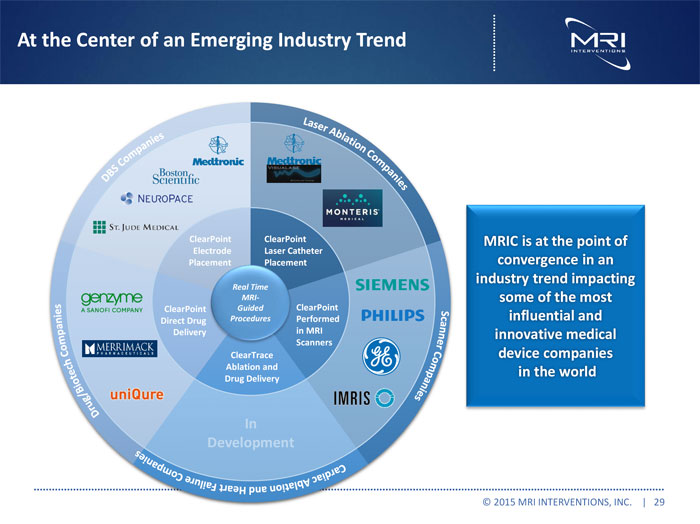
At the Center of an Emerging Industry Trend
ClearPoint ClearPoint MRICisatthepointof
Electrode Laser Catheter
Placement Placement convergenceinan
industrytrendimpacting
Real Time
MRI- someofthemost
ClearPoint Guided ClearPoint
Direct Drug Procedures Performed influentialand
Delivery in MRI innovativemedical
Scanners
ClearTrace devicecompanies
Ablation and in the world Drug Delivery
In Development
c 2015 MRI INTERVENTIONS, INC. | 29

Ticker: MRIC
MRI Interventions, Inc.
Irvine, CA 949.900.6833 mriinterventions.com
Transforming minimally invasive neurosurgery by enabling real-time visualization with MRI
c 2015 MRI INTERVENTIONS, INC. | 30